The so-called wetting is the phenomenon where a gas adsorbed on a solid surface is replaced by a liquid. Substances that enhance this replacement capability are called wetting agents. Wetting is generally divided into three categories: contact wetting, immersion wetting, and spreading wetting. Among them, spreading is the highest standard of wetting, often measured by the spreading coefficient, serving as an indicator of the wetting performance between systems.
In addition, the size of the contact angle is also a criterion for judging the quality of wetting. Surfactants can be used to control the degree of wetting between liquids and solids. In the pesticide industry, some granules and powder formulations for spraying contain a certain amount of surfactants. The purpose is to enhance the adhesion and deposition of the pesticide on the treated surface, increase the release rate and surface area of the active ingredient under conditions of moisture, and improve the effectiveness of disease prevention and treatment.
Application of Surfactants in Agriculture
In the cosmetics industry, as an emulsifier, surfactants are indispensable components in skincare products such as creams, lotions, cleansers, and makeup removers.

The critical micelle concentration (CMC) is the minimum concentration at which surfactant molecules form micelles in a solution. When the concentration exceeds the CMC value, surfactant molecules arrange themselves into structures such as spheres, rods, bundles, layers, or sheets.
Solubilization is a thermodynamic equilibrium process. The lower the CMC and the higher the aggregation number, the greater the solubilization capacity (MAC). Temperature influences solubilization; it affects micelle formation, solubilization of solutes, and the solubility of surfactants. The solubility of ionic surfactants sharply increases with temperature, known as the Krafft point. A higher Krafft point corresponds to a lower critical micelle concentration.
In nonionic surfactants like polyethylene glycol types, as the temperature increases, solubility sharply decreases, resulting in phase separation and cloudiness, known as the cloud point. Longer hydrophobic chains lead to lower cloud points, while longer polyethylene glycol chains lead to higher cloud points.
For nonpolar organic compounds like benzene with low solubility in water, adding surfactants like sodium oleate significantly increases their solubility, a phenomenon known as solubilization. In solubilization, the solubilized benzene is not uniformly dispersed in water but forms micelles with sodium oleate molecules. Various micelles are confirmed to increase in size after solubilization, while the overall solution properties undergo minimal changes.
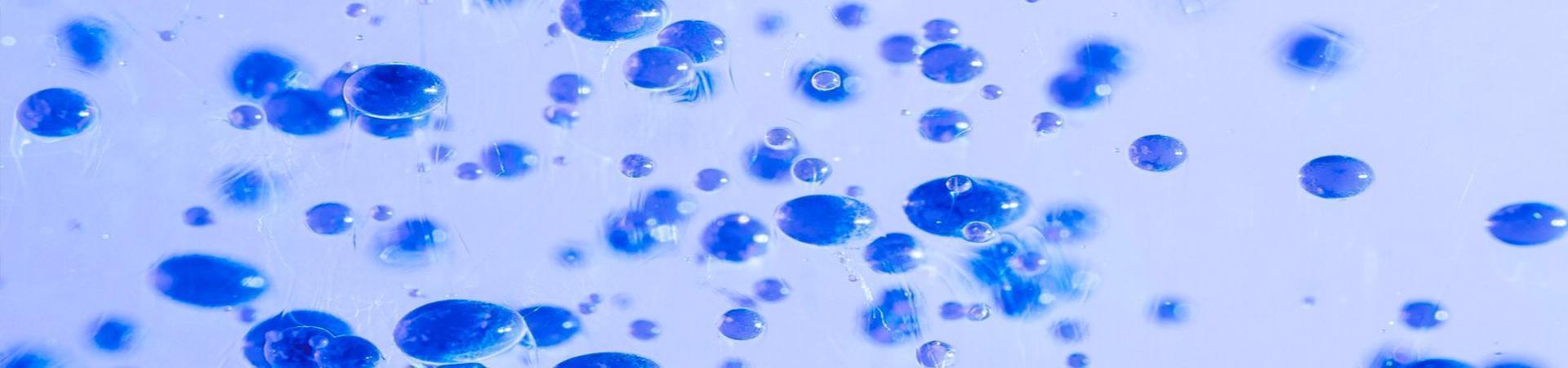
As the concentration of surfactants increases in water, surfactant molecules on the surface form a tightly packed monolayer with oriented alignment, and excess molecules in the bulk phase approach each other in groups of three, forming micelles. The concentration at which micelle formation begins is called the critical micelle concentration.
At this point, solution properties deviate from ideal behavior, and a turning point appears on the concentration vs. surface tension curve. Further increasing surfactant concentration results in no further reduction in surface tension, but the number and size of micelles in the bulk phase continue to increase.
Surfactants, when dispersed in a solution, form micelles when the concentration reaches a certain level. This concentration, known as the critical micelle concentration (CMC), marks the transition to the formation of micelles, and the process is called micellization.
In the process of micellization, as the concentration of surfactants changes in the solution, there is a transition. In extremely dilute solutions, where the surfactant concentration is very low, air and water are in direct contact, and the surface tension of water decreases slightly, approaching the state of pure water. As the surfactant concentration increases slightly, surfactant molecules quickly aggregate at the water surface, reducing the contact between water and air, leading to a rapid decrease in surface tension. Some surfactants start forming small micelles, and with a gradual increase in surfactant concentration, when the surfactant solution reaches saturation adsorption, a tightly packed monolayer of surfactant molecules is formed at the liquid surface. When the concentration reaches the critical micelle concentration (CMC) of the surfactant, the surface tension of the solution drops to its minimum value. Beyond the CMC, although the concentration of the solution increases, the surface tension hardly decreases. At this point, the solution is composed of micelles, and the system gradually transitions to a liquid crystal state.
For ionic surfactants, the micelles formed have a high charge. Due to electrostatic forces, counterions are attracted around the micelles, partially neutralizing the positive and negative charges. However, after the formation of highly charged micelles, the hindrance to the formation of ion clouds by counterions significantly increases. This phenomenon is utilized to adjust the dispersibility of nanoscale particles. The equivalent conductivity of the solution rapidly decreases after reaching the CMC, indicating a change in the system's properties.
The structure of micelles formed by ionic surfactants is spherical, consisting of an inner core, an outer shell, and a diffuse double layer. The inner core is composed of hydrophobic hydrocarbon chains resembling liquid hydrocarbons, with a diameter of approximately 1-2.8 nm. Due to the polar nature of adjacent -CH2- groups, water molecules are present around the core, resulting in a significant amount of permeating water. The -CH2- groups in the core are not exclusively composed of liquid hydrocarbon chains but also form part of the non-liquid outer shell of the micelle.
The outer shell of the micelle, also referred to as the "interface" or surface phase of the micelle-water, consists of the innermost layer of the micelle's double layer, known as the Stern layer (or fixed adsorption layer), with a thickness of approximately 0.2-0.3 nm. The micelle's outer shell includes not only the ionic head and a fixed portion of counterions but also a hydration layer due to ion hydration. The micelle's outer shell is not a smooth surface but rather a "rough" and uneven surface caused by thermal motion of surfactant monomer molecules, leading to fluctuations in the micelle's outer shell.
In a non-aqueous (oily) medium where oil molecules predominate, the hydrophilic groups of surfactants within the micelle form a polar core, while the hydrophobic hydrocarbon chains constitute the outer shell of the micelle. This arrangement results in the formation of reverse micelles, as opposed to conventional micelles formed in water, known as normal micelles. The figure illustrates the model of reverse micelle formation by surfactants in a non-aqueous solution. Reverse micelles have found widespread use in the synthesis and preparation of nanodrug carriers, including the encapsulation of hydrophilic drugs.
The emulsification effect involves the comprehensive affinity of hydrophilic and hydrophobic groups in surfactant molecules towards both oil and water. Empirically, the Hydrophilic-Lipophilic Balance (HLB) values of surfactants are restricted within the range of 0-40, with nonionic types having HLB values within 0-20.
The mixed-additivity formula for calculating HLB is expressed as: HLB = (HLB_a * W_a + HLB_b * W_b) / (W_a + W_b). The theoretical calculation of HLB is given by: HLB = ∑(hydrophilic group HLB values) + ∑(lipophilic group HLB values) - 7. The HLB ranges for different emulsifiers are as follows: HLB 3-8 for W/O type emulsifiers (e.g., Tween; monovalent soaps), and HLB 8-16 for O/W type emulsifiers (e.g., Span; divalent soaps).
A coarse dispersion system formed by dispersing one or more liquids, in droplet form with a diameter greater than 10-7m, in another immiscible liquid is referred to as an emulsion. To maintain its stability, an emulsifying agent must be added. Depending on the structure of the emulsifying agent, emulsions can be classified as water-in-oil (O/W), where water is the continuous phase, or oil-in-water (W/O), where oil is the continuous phase. Sometimes, to break the emulsion, another type of surfactant known as a demulsifier is added to separate the dispersed phase and the dispersing medium in the emulsion. For example, in crude oil, a demulsifier is added to separate oil and water.
Surfactants find widespread applications in the pharmaceutical industry. In pharmaceutical formulations, surfactants play a crucial role in solubilizing many poorly soluble drugs, such as volatile lipid-soluble cellulose and steroidal hormones. The use of surfactants enhances solubility, leading to the formation of transparent solutions and increased drug concentrations. In pharmaceutical preparation processes, surfactants are indispensable as emulsifiers, wetting agents, suspending agents, foaming agents, and defoaming agents.

The term "foam" refers to a collection of gas surrounded by a liquid film. Some surfactants, when combined with water, can form films with sufficient strength to enclose air and create foam. This foaming effect is utilized in processes such as froth flotation in ore beneficiation, foam firefighting, and detergent-based stain removal. Surfactants that promote foaming are referred to as foaming agents.
On the other hand, there are situations where defoaming agents are necessary. In processes like sugar refining or traditional Chinese medicine preparation, excessive foam formation may occur. In such cases, specific surfactants are added to reduce the strength of the liquid film, eliminate bubbles, and prevent accidents by acting as defoaming agents.
In the pesticide industry, wettable powders, emulsifiable concentrates, and concentrated emulsions all require a certain amount of surfactants. For example, in wettable powders, the active ingredients are often organic compounds with hydrophobic properties. Only under the conditions of surfactant presence, reducing the surface tension of water, can the pesticide particles be wetted by water, forming a water suspension.

Surfactants are employed in the froth flotation of ores to enhance the flotation process. By stirring and introducing air from the bottom of the tank, bubbles containing effective mineral fines gather on the surface. These bubbles are then collected and deaerated through concentration, achieving the goal of mineral enrichment. The sediments such as sand and rocks without minerals remain at the bottom of the tank and are periodically removed. When the surface of mineral sands is covered by a capturing agent by 5%, it imparts hydrophobicity to the surface. This hydrophobic surface attaches to the bubbles, rising to the liquid surface for easy collection. Choosing an appropriate capturing agent ensures that its hydrophilic groups selectively bind to the surface of mineral sands, while the hydrophobic groups face the water.
In the pharmaceutical industry, surfactants are employed as sterilizing agents and disinfectants. Their antimicrobial and disinfecting effects stem from their strong interaction with bacterial biofilm proteins, leading to denaturation or loss of function. These disinfectants exhibit significant solubility in water, and depending on the concentration, they can be utilized for preoperative skin disinfection, wound or mucosal disinfection, instrument sterilization, and environmental disinfection.

The removal of grease stains is a complex process that involves wetting, foaming, and other related actions mentioned above. Various auxiliary ingredients are typically added to detergents to enhance their wetting effects on the object being cleaned, while also providing functions such as foaming, whitening, and preventing recontamination of the cleaned surface. The primary component, surfactant, plays a crucial role in the degreasing process: water has high surface tension and poor wetting properties for oil stains, making it challenging to wash away grease. When surfactants are added, hydrophobic groups orient towards the fabric surface and adhere to the dirt, gradually detaching it from the surface. The dirt is then suspended in water or removed as foam rises to the water surface, leaving the clean surface occupied by surfactant molecules.
It is important to note that the effectiveness of surfactants is not solely due to a specific action; in many cases, multiple factors work in concert. In the papermaking industry, for instance, surfactants are utilized as cooking agents, ink removal agents for waste paper, sizing agents, resin barrier control agents, defoaming agents, softeners, antistatic agents, scale inhibitors, softening agents, degreasers, bactericides, algaecides, corrosion inhibitors, and more.