Commonly used surfactants are often low-molecular-weight compounds with molecular weights in the hundreds. With the in-depth research in various hot fields such as enhanced oil recovery (EOR), drug carriers and controlled release, biomimicry, polymer LB films, medical high-polymer materials (anticoagulation), emulsion polymerization, the requirements for surfactants are becoming more diverse and high-performance. High-molecular-weight compounds with surfactant properties have now become a focal point of attention.Substances with molecular weights above several thousand and possessing surfactant properties are commonly referred to as polymeric surfactants. Similar to conventional surfactants, there is currently no standardized classification for polymeric surfactants.
1.Classification of Polymeric Surfactants:
Based on the formation of micelles in solution, polymeric surfactants can be classified into polymer soaps and water-soluble polymeric surfactants.
According to the classification of low molecular weight surfactants based on their ionic nature in water, they can be categorized as anionic, cationic,amphoteric, and nonionic.
2.Structure and Performance of Polymeric Surfactants:
Block-type surfactants,
Comb-type surfactants,
3.Applications of Polymeric Surfactants:
Applications in the pharmaceutical industry
Applications in the petroleum industry
Applications in the textile dyeing and printing industry
Applications in the papermaking industry
Depending on whether they form micelles in solution, they can be further divided into soap-like and water-soluble polymeric surfactants.
1.1 Polysoaps
The majority of polysoaps carry a charge, resembling polyelectrolytes. In fact, most polysoaps are products of hydrophobic modification of polyelectrolytes and are typically insoluble in water. Currently synthesized polysoaps include the following types (where R represents a long-chain alkyl group in the formulas):
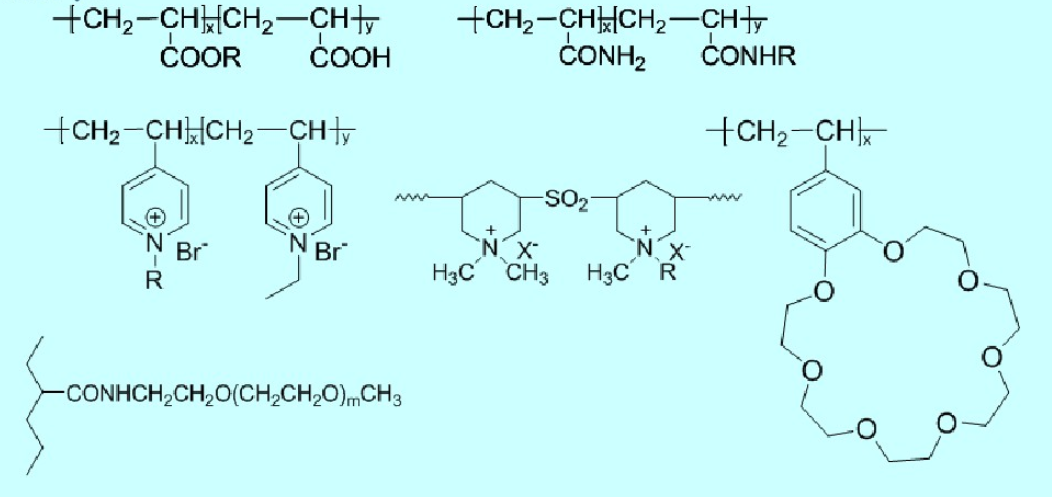
1.2 Water-Soluble Polymeric Surfactants
Polymeric surfactants that do not form micelles in solution are generally referred to as water-soluble polymeric surfactants. They are categorized into three main classes based on their sources: natural, semi-synthetic, and synthetic polymeric surfactants.
Natural polymers include various common substances such as gum, starch, microbial fermentation polysaccharides, and others.
Semi-synthetic polymers are polymers derived from chemical modifications of substances like starch, cellulose, and proteins, resulting in various polymers such as cationic starch and methyl cellulose.
Synthetic polymers are derived from the polymerization of monomers obtained through petrochemical processes. Examples include derivatives of polyacrylamide and polyacrylic acid.
Based on their ionic nature in water, polymeric surfactants can be categorized into anionic surfactants , cationic surfactants , amphoteric surfactants, and nonionic surfactants types.
Anionic Polymeric Surfactants
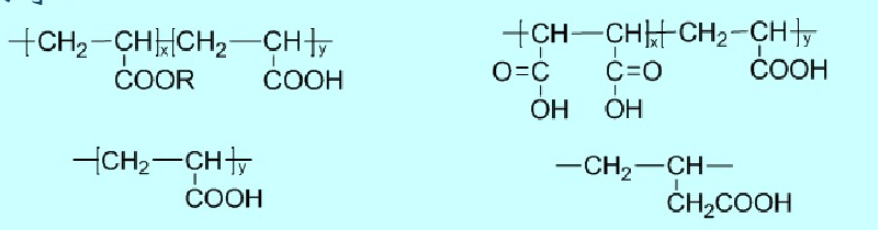
(1) Carboxylic Acid Type
Typical polymers include polyacrylic acid and its copolymers, butenoic acid and its copolymers, copolymers of acrylic acid and maleic anhydride, and their partially saponified derivatives.
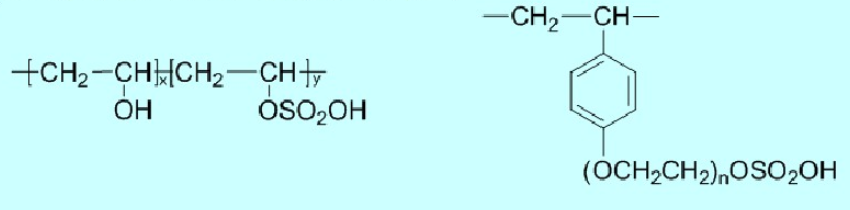
(2) Sulfate Ester Type
Typical polymers include:
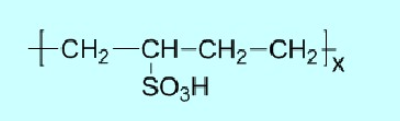
(3) Sulfonic Acid Type
This category includes partially sulfonated polystyrene, benzene sulfonic acid-formaldehyde condensates, naphthalene sulfonic acid-formaldehyde condensates, sulfonated polybutadiene, and lignosulfonates, which are also a type of sulfonic acid-type polymeric surfactant. Typical sulfonic acid-type polymeric surfactants include:
Cationic Polymeric Surfactants
(1) Amine Salts or Polyamines
Examples include polyethylene imine, polyethyleneimine-based pyrrolidone, poly(maleimide) and its derivatives, etc. Typical polymers include:
(2) Quaternary Ammonium Salts
Examples include quaternized polyacrylamide, polyvinylpyridine salts, and polydiallylamine epichlorohydrin, etc. Quaternary ammonium polymeric surfactants exhibit cationic properties in acidic, neutral, and alkaline aqueous media. Representative products include:
(3) Amphiphilic Polymeric Surfactants
Main varieties include copolymers of acrylic acid and vinylpyrrolidone, copolymers of acrylic acid and cationic acrylate, amphoteric polyacrylamide, etc. Examples include:
(4)Nonionic Polymeric Surfactants
Main varieties include polyvinyl alcohol and its partially esterified or aldehyde-condensed products, such as modified polyacrylamide, copolymers of maleic anhydride, polypropylene acrylate, polyether, polyethylene oxide-propylene oxide copolymer, water-soluble phenolic resins, amino resins, etc.
The surface activity of polymeric surfactants depends on the molecular conformation in solution, which is closely related to the amphiphilic chemical structure, composition, and relative molecular weight of the polymer.
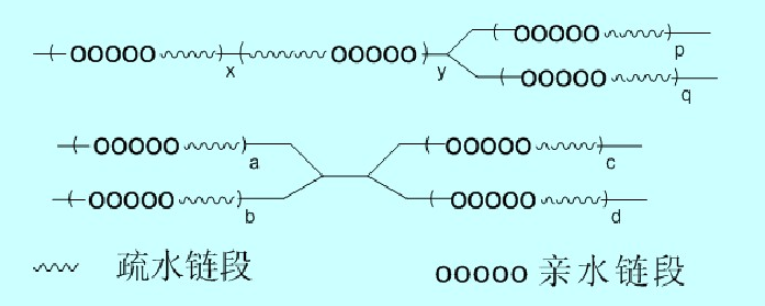
Block Copolymeric Surfactants
In block copolymers, hydrophobic segments are distributed along the polymer backbone. An appropriate sequence length of hydrophobic and hydrophilic portions will effectively prevent self-aggregation of the hydrophobic segments (formation of single-molecule micelles) or intermolecular aggregation (multimolecular aggregation).
Comb-like Surfactants
Comb-like surfactants have advantages such as easy preparation and diverse varieties. These surfactants are obtained by homopolymerization or copolymerization of both amphoteric and amphiphilic monomers. Depending on the position of hydrophobic and hydrophilic groups, they exhibit various branched chemical structures.
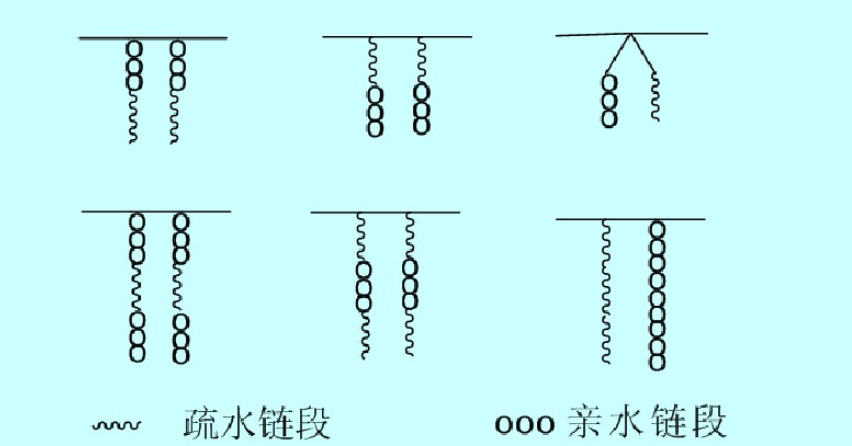
Due to the presence of hydrophilic groups on the side chains, the aggregation of hydrophobic segments is hindered. Even in the formed micelles, the internal structure is relatively loose compared to micelles with tightly packed cores, allowing for a considerable amount of water molecules. Therefore, it can exhibit high surface activity. Additionally, due to the configuration, the amphiphilic side chains hinder the aggregation of the hydrophobic main chain composed of methylene and methyl groups, preventing it from participating in interfacial adsorption.
Studies have indicated that, under the condition of maintaining solubility, any factor increasing the molecular chain rigidity is favorable for the expansion of macromolecules in the solution, potentially enhancing the surface activity of polymers.
In the Pharmaceutical Industry
Due to the excellent surface activity of block and graft polymeric surfactants, they find widespread applications in the pharmaceutical industry. They can be utilized as drug carriers, emulsifiers, dispersion solvents, wetting agents, and more. Additionally, polymeric surfactants serve as phase-transfer catalysts in pharmaceutical synthesis and have extensive applications in drug analysis.
Application in the Petroleum Industry
Due to the presence of solid paraffin in extracted crude oil, the poor fluidity of crude oil poses challenges in the production, storage, and processing of this easily gelling and highly viscous material. This issue can be addressed by adding pour point depressants or flow improvers. The dispersibility of oil-soluble polymeric surfactants can further enhance the effectiveness of flow improvers, preventing the precipitation of paraffin in fuel oil during transportation and storage.
Application in the Textile Printing and Dyeing Industry
Polyether-type polymeric surfactants are commonly used as low-foaming detergents, emulsifiers, dispersants, defoamers, antistatic agents, wetting agents, and dyeing agents in the textile printing and dyeing industry. Polymeric compounds such as polyvinyl alcohol find extensive use as thickeners and protective colloids in the preparation of emulsion-type dyeing auxiliaries. Cellulose derivatives like carboxymethyl cellulose are employed in detergents as re-deposition inhibitors. Lignosulfonates, phenol-formaldehyde condensate sulfonates, and other sulfonated compounds serve as dispersants for insoluble dyes.

Application in the Papermaking Industry
Due to the unique and significant role of high-molecular-weight surfactants in improving paper performance and enhancing paper machine efficiency, they have increasingly gained attention from professionals in the papermaking industry. Studies have indicated that the preparation of maleic acid monoesters using polyethylene glycol and maleic anhydride with different molecular weights, followed by copolymerization with acrylic acid to produce maleic acid monoester/acrylic acid copolymers, demonstrates remarkable ink removal effects.
